“The good physician treats the disease;
the great physician treats the patient who has the disease.”
Sir William Osler, Father of Modern Medicine
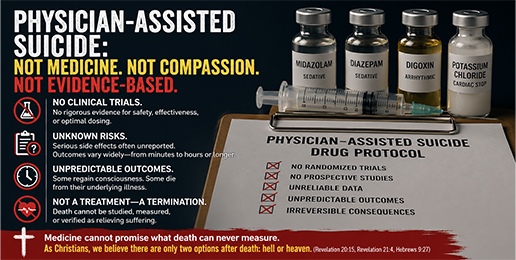
Nothing in this article denies the sincere compassion many clinicians and families feel. The issue is whether the drug protocols and oversight meet the normal evidence-based standards we would demand for any medical intervention, especially one that aims at death. At the outset, it is important to understand that there are no randomized controlled trials or prospective clinical trials testing which drugs should be used for Physician Assisted Suicide (PAS). There are no published studies that systematically evaluate whether these drug combinations are safe, reliable, or predictable, or that carefully study their side effects, optimal dosing, or how the body processes them. In other areas of medicine, such studies are required before drugs are approved or widely used. That is not the case here.
Current recommendations for PAS are based mainly on observational reports and surveillance data, most of which come from Oregon. These reports are not controlled clinical trials. They are not designed to rigorously test safety, effectiveness, or predictability, and they rely largely on accumulated experience rather than high-quality medical evidence. For this reason, the drug combinations currently used for physician assisted suicide would not meet the evidence based standards normally required for therapeutic approval. The available information is incomplete and inconsistently reported.
The most commonly used drug combination includes medications intended to cause deep sedation, suppress breathing, and disrupt heart rhythm. Several of these drugs overlap in their effects. This approach is used not because it is precise or well studied, but because absorption and clinical effects can vary widely from person to person.
Reported side effects include nausea, vomiting, low blood pressure, deep sleep progressing to coma, choking or aspiration into the lungs, regurgitation of medication, agitation, and, in rare cases, seizures. Information about these side effects is incomplete, largely because a physician is usually not present at the time the drugs are taken or when death occurs, and many reports list complications as unknown.
The time it takes for death to occur varies widely, ranging from minutes to many hours. In some reported cases, death has taken longer than a full day. In Oregon’s reported cases with known time-to-death data, approximately 5 to 7 percent took more than six hours. Rarely, Oregon has recorded cases in which individuals regained consciousness after ingestion and later died from their underlying illness rather than from taking additional medication. Even in these cases, details are limited and often not recorded by medical professionals.
Although death is frequently studied as an endpoint in medicine, PAS does not allow for meaningful assessment of patient experience after ingestion. In other areas of medicine, patient-reported outcomes are essential for understanding distress, suffering, or unexpected harm. With PAS, this information is largely unavailable due to limited observation and the absence of systematic follow-up.
Outcome
The ultimate outcome of PAS is death, and this creates a fundamental problem because the claim that death relieves suffering cannot be studied, measured, or verified. Medicine cannot promise that death relieves suffering, because once a patient is dead there is no way to assess subjective benefit, harm, or quality of outcome. Unlike other medical interventions, death offers no patient-reported endpoints, no meaningful follow-up, and no therapeutic mechanism as traditionally defined in medicine beyond ending the patient’s life. From an objective medical standpoint, choosing death is not a treatment but a termination of care, with consequences that neither physicians nor patients can fully understand, predict, or control. As Christians, we believe there are only two options after death: hell or heaven. (Revelation 20:15, Revelation 21:4, Hebrews 9:27) .





















